Products You May Like
Scientists have built the tiniest antenna ever made – just five nanometers in length. Unlike its much larger counterparts we’re all familiar with, this minuscule thing isn’t made to transmit radio waves, but to glean the secrets of ever-changing proteins.
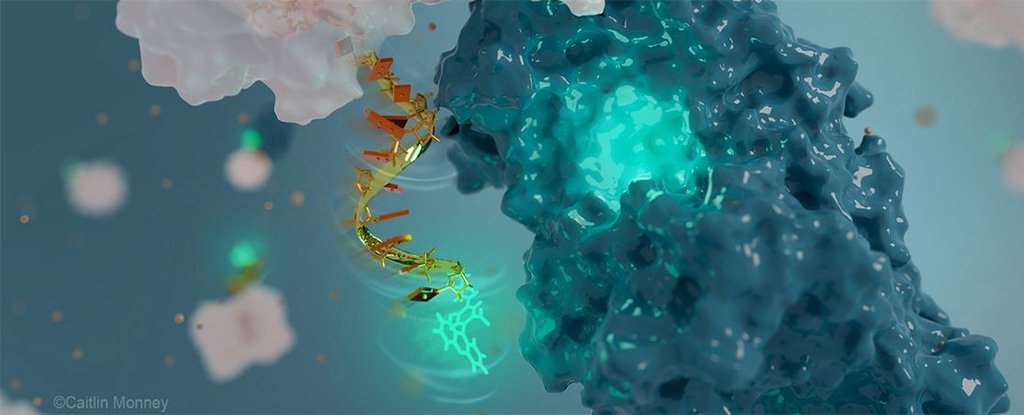
The nanoantenna is made from DNA, the molecules carrying genetic instructions that are around 20,000 times smaller than a human hair. It’s also fluorescent, which means it uses light signals to record and report back information.
And those light signals can be used to study the movement and change of proteins in real time.
Part of the innovation with this particular antenna is the way in which the receiver part of it is also used to sense the molecular surface of the protein it’s studying. That results in a distinct signal when the protein is fulfilling its biological function.
“Like a two-way radio that can both receive and transmit radio waves, the fluorescent nanoantenna receives light in one color, or wavelength, and depending on the protein movement it senses, then transmits light back in another color, which we can detect,” says chemist Alexis Vallée-Bélisle, from the Université de Montréal (UdeM) in Canada.
Specifically, the job of the antenna is to measure the structural changes in proteins over time. Proteins are large, complex molecules that carry out all kinds of essential tasks in the body, from supporting the immune system to regulating the function of organs.
However, as proteins rush about doing their jobs, they undergo constant changes in structure, transitioning from state to state in a highly complex process scientists call protein dynamics. And we don’t really have good tools to track these protein dynamics in action.
“Experimental study of protein transient states remains a major challenge because high-structural-resolution techniques, including nuclear magnetic resonance and X-ray crystallography, often cannot be directly applied to study short-lived protein states,” the team explains in their paper.
The latest DNA synthesizing technology – some 40 years in development – is able to produce bespoke nanostructures of different lengths and flexibilities, optimized to fulfil their required functions.
One advantage that this super-small DNA antenna has over other analysis techniques is that it’s able to capture very short-lived protein states. That, the researchers say, means there are plenty of potential applications here, in both biochemistry and nanotechnology more generally.
“For example, we were able to detect, in real time and for the first time, the function of the enzyme alkaline phosphatase with a variety of biological molecules and drugs,” says chemist Scott Harroun, from UdeM. “This enzyme has been implicated in many diseases, including various cancers and intestinal inflammation.”
While exploring “the universality” of their design, the team successfully tested their antenna with three different model proteins – streptavidin, alkaline phosphatase and Protein G – but there’s potentially much more to come, and one of the advantages of the new antenna is its versatility.
“Nanoantennas can be used to monitor distinct biomolecular mechanisms in real time, including small and large conformational changes – in principle, any event that can affect the dye’s fluorescence emission,” the team writes in their paper.
DNA is becoming more and more popular as a building block that we can synthesize and manipulate to create nanostructures like the antenna in this study. DNA chemistry is relatively simple to program, and easy to use once programmed.
The researchers are now looking to create a commercial startup so that the nanoantenna technology can be practically packaged and used by others, whether that’s pharmaceutical organizations or other research teams.
“Perhaps what we are most excited by is the realization that many labs around the world, equipped with a conventional spectrofluorometer, could readily employ these nanoantennas to study their favorite protein, such as to identify new drugs or to develop new nanotechnologies,” says Vallée-Bélisle.
The research has been published in Nature Methods.